RNA Based Therapeutics
Over the past decades RNA based therapeutics gained importance. Especially in diseases with genetic causes, oligonucleotide chains offer a promising therapy approach - often the first ever. Currently there are two main approaches used to target RNA: double stranded RNA-mediated interference (RNAi) and antisense oligonucleotides (ASO). Both approaches are currently in clinical trials for targeting of RNAs involved in various diseases, such as cancer and neurodegenerative diseases. antibodies-online can support you in the development of RNA based therapeutics. We offer ModDetect™ panels, which are designed to facilitate the detection of specific chemical modifications independent of the sequence or location of the modification and can be used to evaluate a variety of RNA Tx modalities and nucleic acid structures. Discover the panels directly or learn more below.
Challenges in Therapeutic Production
Due to its sequence specificity, an oligo therapeutic can in principle be used against any mRNA and thus against any disease treatable by gene knockdown. However, the use of oligos also comes with potential obstacles, such as the comparatively rapid degradation of RNA in the bloodstream and the targeted uptake of the therapeutic — preferably in diseased cells or tissues of an organism.
The evolution of the medicinal chemistry of oligonucleotides has been critical to the steadily improving performance of ASOs in the clinic. ASOs are oligomeric and comprised of nucleotide analogs. Because ASOs can be designed to work through a variety of post-RNA-binding mechanisms, numerous designs have been evaluated. As new molecular mechanisms of action are identified and new insights into the molecular mechanisms of distribution, cellular uptake and subcellular distributions, and various toxicities are reported, the designs are becoming progressively more complex.
Product Spotlight: Rockland Moddetect® Panels
These oligonucleotides often contain one or more structural modifications that increase their stability, uptake and effectiveness but may increase the likelihood of toxicity. Balancing the level of modification with the effectiveness of the therapeutic candidate often results in large oligonucleotide libraries that lead to increased time required for screening and associated reagent expense. Traditionally, ISH-type assays have been the only method of detecting and localizing oligonucleotides in cells and tissues for nucleic acid therapy development. However, with the inability for ISH to bind and detect short oligo therapeutic drug sequences and because it requires unique probes to each oligonucleotide candidate, the process for probe development and candidate triage continues to require significant time and excessive budgets.
Rockland's ModDetect™ panels are ready-to-use, immunoassay-based alternatives that provide robust detection and localization of oligonucleotide therapeutic drug delivery, thereby facilitating the collection of ADME analytical data for regulatory approval. The universal nature and unique specificity and sensitivity of these reagents eliminates the need for unique probes and accelerates candidate triage for a cost-effective, low-risk solution that can save 9-12 months in drug development.
Available Moddetect® Panels
ModDetect® Phosphorothioate Panel
ABIN7675637

ModDetect® Phosphorothioate Biotinylated Panel
ABIN7675638

ModDetect® 2'-O-Methyl (2'OMe) Panel
ABIN7675641
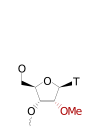
ModDetect® 2-Methoxyethyl (2'MOE) Panel
ABIN7675639
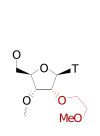
ModDetect® 2-Methoxyethyl (2'MOE) Biotinylated Panel
ABIN7675640

ModDetect™ Phosphorothioate (PS) Panel
The ModDetect™ Phosphorothioate (PS) Panel enables antibody-based detection of PS-modified oligonucleotides commonly used in therapeutic development. PS modifications improve nuclease resistance and pharmacokinetic properties but are difficult to assess using sequence-specific analytical tools. This panel recognizes the PS chemical linkage itself, allowing sequence-independent analysis across diverse oligonucleotide designs. The antibodies are compatible with standard immunoassays, including ELISA, immunofluorescence, and tissue-based applications. These reagents support biodistribution, localization, and quantitative studies during research and preclinical development. Learn more
ModDetect™ Phosphorothioate (PS) Biotinylated Panel
The ModDetect™ Phosphorothioate (PS) Biotinylated Panel is designed for immunoassay workflows requiring biotin-mediated capture or detection. It provides modification-specific recognition of PS linkages while enabling flexible assay configurations. Biotinylation supports use in sandwich ELISAs, plate-based capture assays, and streptavidin-driven formats. This panel enables sequence-independent detection of PS-modified oligonucleotides across multiple therapeutic modalities. It is well suited for quantitative and comparative bioanalytical applications in research and preclinical studies. Learn more
ModDetect™ 2′-O-Methoxyethyl (MOE) Panel
The ModDetect™ 2′-O-Methoxyethyl (MOE) Panel enables antibody-based detection of MOE-modified oligonucleotides used to enhance stability and tolerability. MOE modifications are frequently incorporated into antisense and RNA-based therapeutics but are challenging to evaluate with conventional assays. This panel targets the MOE chemical feature directly, allowing sequence-independent analysis across oligonucleotide designs. The antibodies are compatible with ELISA, immunofluorescence, and tissue-based assays. These reagents support biodistribution, localization, and quantitative assessment in research and preclinical workflows. Learn more
ModDetect™ 2′-O-Methoxyethyl (MOE) Biotinylated Panel
The ModDetect™ 2′-O-Methoxyethyl (MOE) Biotinylated Panel supports immunoassay formats that require biotin-enabled capture or detection. It retains the same modification-specific recognition as the unconjugated MOE panel while expanding assay design flexibility. Biotinylation allows integration into sandwich ELISAs and streptavidin-based platforms. This panel enables sequence-independent detection of MOE-modified oligonucleotides across multiple therapeutic modalities. It is ideal for quantitative and comparative studies in research and preclinical development. Learn more
ModDetect™ 2′-O-Methyl (OMe) Panel
The ModDetect™ 2′-O-Methyl (OMe) Panel enables antibody-based detection of OMe-modified oligonucleotides across a range of nucleic acid modalities. OMe modifications are commonly used to improve stability and reduce immune activation but are not easily analyzed using sequence-dependent methods. This panel detects the OMe chemical modification itself, enabling sequence-independent evaluation. The antibodies are compatible with standard immunoassays, including ELISA, immunofluorescence, and tissue-based assays. These reagents support studies of oligonucleotide distribution, localization, and comparative performance. Learn more
Oligonucleotide Medicinal Chemistry: Modifications and their Advantages
The phosphorothioate (PS) modification is broadly used in all major classes of ASOs and all chemically modified siRNAs. The replacement of one nonbridging oxygen with a sulfur alters the physicochemical characteristics of the phosphate in important ways. Because the sulfur atom is twice as large as the oxygen atom, the charge distribution, bond angles, and stretching of PS links differ substantially from phosphodiester (PO) linkages. The sulfur substitution spreads the charge and makes the phosphate more “lipophilic,” thereby facilitating binding to proteins. Generally, for proteins that require PS moieties to bind ASOs, the minimum number of this modification needed to support meaningful protein interactions is 10. The enhanced protein binding enabled by PS substitutions is critical as protein binding of ss PS ASOs plays crucial roles in the absorption, distribution, cellular uptake, intracellular distribution, activity, and toxicity of PS ASOs.
Modifications at the 2′ position of the ribose ring are commonly used to help increase oligonucleotide stability and improve resistance to nuclease activity in vivo. RNA oligonucleotides synthesized using 2′-MOE modifications, known as phosphoramidites, have shown to be more nuclease resistant, with lower toxicity, and slightly increased hybridization affinities, making them well suited for therapeutic in vivo applications, such as ASO, siRNA, and aptamers. 2'-O-methylnucleotides offer advantages due to their kinetic and melting properties. 2'-O-Methyl oligoribonucleotide probes bind to RNA targets faster and with much higher melting temperatures (Tm) at various probe length. Because of their greatly enhanced Tm when bound to RNA, 2'-O-methyl oligoribonucleotide probes can efficiently bind to double-stranded regions of structured RNA molecules. The increased Tm, faster kinetics of hybridization, ability to bind to structured targets and increased specificity of 2'-O-methyl oligoribonucleotide probes render them superior to corresponding 2'-deoxy oligoribonucleotides for use in assays that detect RNA targets.
PS: Phosphorothioate

2’-OMe: 2'-O-methylated Nucleosides

2’-MOE: 2-Methoxyethyl

2’-LNA: Locked Nucleic Acid

2′-MOE-modified oligonucleotides have demonstrated that the oligonucleotides exhibit a tissue distribution similar to phosphorothioate oligodeoxynucleotides (PS ODNs) and decreases toxicities compared to PS ODNs. Additionally, 2ʹ-MOE substitution significantly reduces proinflammatory effects.
Therapeutic viability of siRNA gene silencing is dependent on improvements in molecule bio-stability, specificity and delivery, which can be overcome by modifying of siRNA with Locked Nucleic Acid (LNA), a nucleic acid analogue with unprecedented binding affinity. LNA provides excellent specificity toward complementary RNA and DNA oligonucleotides. Incorporating LNA substantially enhances serum half-life of siRNA's, which is a key requirement for therapeutic use. Moreover LNA is compatible with the intracellular siRNA machinery and can be used to reduce undesired, sequence-related off-target effects. The remarkable properties of LNA have led to applications within various gene silencing strategies both in vitro and in vivo.
Case Study: Antisense Oligonucleotide Silencing of FUS
Fused in sarcoma (FUS) is an RNA-binding protein that is genetically and pathologically associated with rare and aggressive forms of amyotrophic lateral sclerosis (ALS) and frontotemporal dementia (FTD). Korobeynikov et al researched on ION363, a non-allele-specific FUS antisense oligonucleotide. ION363 efficiently silences FUS and reduces postnatal levels of FUS protein in the brain and spinal cord, delaying motor neuron degeneration. In mouse genetic and human clinical studies, evidence is provided in support of FUS silencing as a therapeutic strategy in FUS-dependent ALS and FTD.
Reliable Detection of Modified Oligos
In production processes of therapeutic agents, rigorous and consistent quality control is key. antibodies-online, together with Rockland Immunochemicals, is able to support your development quality control processes. Rockland’s ModDetect™ Panels are speciality reagent panels developed to detect oligo modifications independent of the sequence or location of the modification. This makes the ModDetect™ Panels useful for oligonucleotide therapeutic development, mRNA vaccine development, or research of genetic diseases or gene expression.
Get in touch with our experts at antibodies-online and Rockland to discuss antibodies against oligo modifications customized for your needs.
Related Products and Resources
References
- : "Control of phosphorothioate stereochemistry substantially increases the efficacy of antisense oligonucleotides." in: Nature biotechnology, Vol. 35, Issue 9, pp. 845-851, (2017) (PubMed).
- : "Cellular uptake and trafficking of antisense oligonucleotides." in: Nature biotechnology, Vol. 35, Issue 3, pp. 230-237, (2017) (PubMed).
- : "Short antisense oligonucleotides alleviate the pleiotropic toxicity of RNA harboring expanded CGG repeats." in: Nature communications, Vol. 12, Issue 1, pp. 1265, (2021) (PubMed).
- : "Antisense technology: A review." in: The Journal of biological chemistry, Vol. 296, pp. 100416, (2021) (PubMed).
- : "Antisense technology: an overview and prospectus." in: Nature reviews. Drug discovery, Vol. 20, Issue 6, pp. 427-453, (2021) (PubMed).
- : "Advantages of 2'-O-methyl oligoribonucleotide probes for detecting RNA targets." in: Nucleic acids research, Vol. 26, Issue 9, pp. 2224-9, (1998) (PubMed).
- : "Locked nucleic acid (LNA) mediated improvements in siRNA stability and functionality." in: Nucleic acids research, Vol. 33, Issue 1, pp. 439-47, (2005) (PubMed).
- : "RNA therapeutics: RNAi and antisense mechanisms and clinical applications." in: Postdoc journal : a journal of postdoctoral research and postdoctoral affairs, Vol. 4, Issue 7, pp. 35-50, (2016) (PubMed).
- : "Antisense oligonucleotide silencing of FUS expression as a therapeutic approach in amyotrophic lateral sclerosis." in: Nature medicine, Vol. 28, Issue 1, pp. 104-116, (2022) (PubMed).
- : "Antibody-Oligonucleotide Conjugates as Therapeutic, Imaging, and Detection Agents." in: Bioconjugate chemistry, Vol. 30, Issue 10, pp. 2483-2501, (2020) (PubMed).

Creative mind of antibodies-online with a keen eye for details. Proficient in the field of life-science with a passion for plant biotechnology and clinical study design. Responsible for illustrated and written content at antibodies-online as well as supervision of the antibodies-online scholarship program.
前往作者页面



