DCL4 抗体 (N-Term)
Our Local Distributor
北京 101111
Quick Overview for DCL4 抗体 (N-Term) (ABIN2441994)
抗原
适用
宿主
克隆类型
应用范围
-
-
抗原表位
- N-Term
-
纯化方法
- Affinity purification
-
免疫原
- N-terminal region of arabidopsis thaliana DCL4 (At5g20320)
-
-
-
应用备注
- Optimal working dilution should be determined by the investigator.
-
限制
- 仅限研究用
-
-
- by
- Molecular Plant Sciences, University of Edinburgh
- No.
- #100563
- 日期
- 2017.03.28
- 抗原
- DCL4
- Lot Number
- 1472
- Method validated
- Western Blotting
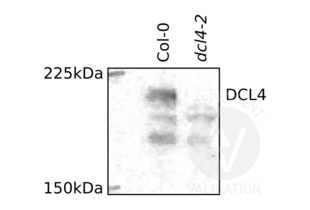
- Positive Control
- Arabidopsis thaliana Col-0
- Negative Control
- Arabidopsis thaliana dcl4-2 DCL4 null mutant
- Notes
- The DCL4 (N2) antibody ABIN2441994 reveals a predominant protein band of the expected molecular weight of DCL4 antigen in lysates of wild type (Col-0) plant cells. This band is not observed in the negative control, using plants of the dcl4-2 null mutant genotype.
- Primary Antibody
- ABIN2441994
- Secondary Antibody
- Anti-Rabbit IgG (H+L) peroxidase conjugated secondary antibody (Thermo Fisher, Catalogue 31466, lot RB230254)
- Full Protocol
- Grow Arabidopsis Col-0 and dcl4-2 null mutant plants in Levington F2+S professional growth compost (Everris LTD product 92150436, lot AF2S75LE) in Panasonic/SANYO growth chambers set to 21°C constant temperature with a 16h light, 8h dark photoperiod.
- Grow plants for 28d.
- Weigh 100mg of leaf tissue in 1.5 ml microcentrifuge tubes containing 2 metal ball bearings.
- Freeze leaf tissue the tubes by submersion in liquid nitrogen.
- Lyze plant tissue in a TissueLyser II (Qiagen, 85300, lot 1212120617C) for 1min at 30Hz.
- Extract total protein by adding 100µl of protein extraction buffer (20mM Tris pH7.5, 5mM MgCl2, 300mM NaCl, 5mM DTT, 0.1% NP40) with 1x cOmplete protease inhibitor cocktail (Roche, 11697498001, lot 11140920) to 100mg of the lyzed plant tissue. Vortex samples for 20s immediately after addition of the protein extraction buffer.
- Centrifuge the crude protein extract at 13,000rpm at 4°C for 1min.
- Transfer the supernatant to a clean microcentrifuge tube and discard the original tube containing the pelleted plant material.
- Use 5µl of the protein extract to determine the protein concentration using a Bradford assay (Sigma, Catalogue B6916, lot SLBL4590V).
- Mix 25µg total plant protein (in 13µl) with 2µl of 10x Bolt LDS sample loading buffer (Novex, B0007, lot 1691095) and 2µl of 10x Bolt sample reducing agent (Novex, B0009, lot 1692508).
- Denature samples in a dry heat block at 70°C for 10min, and immediately load them onto a pre-cast 7% tris-acetate NuPAGE gel (Novex, EA03555BOX, lot 17021432).
- Separate proteins at a constant 165V for 5h at 25°C alongside Amersham full-range rainbow molecular weight marker (GE Healthcare, RPN800E, lot 9661354).
- Wash the gel for 5min in 20% ethanol and soak it for 1min in transfer buffer (Novex, BT0006 & BT0005, lot 1771634).
- Transfer proteins to a 0.45µm nitrocellulose membrane (Bio-Rad, Catalogue 1620115) ON at 4°C and a constant 15V using a Novex miniblot module (Novex, Catalogue B1000).
- Block the membrane in TBSTT (20mM Tris (pH7.5), 150mM NaCl, 0.25% (v/v) Tween-20, 0.1% (v/v) Triton-X for 1h at 25°C.
- Incubate with primary Dicer-Like Protein 4 (DCL4) (N-Term) antibody (antibodies-online, ABIN2441994, lot 1472) diluted 1:2000 in TBSTT for 1h at 25°C.
- Wash the membrane 6x 5min with 5% milk TBSTT.
- Incubate the membrane with Pierce Goat Anti-Rabbit IgG (H+L) peroxidase conjugated secondary antibody (Thermo Fisher, Catalogue 31466, lot RB230254) diluted 1:10000 in 5% milk TBSTT for 1h at 25°C.
- Wash the membrane 6x 10min with 5% milk TBSTT.
- Incubate the membrane for 2min at 25°C in Pierce Western blotting substrate solution containing equal volumes of ECL reagents A/B (Thermo Scientific, Catalogue 32106, lot QG220709B) and SuperSignal ECL reagents A/B (Thermo Scientific, Catalogue 37071, lot RF232237); remove excess Western blotting substrate.
- The membrane was exposed to an X-Ray film (Thermo Scientific, Catalogue 34089) to determine protein abundance.
- Experimental Notes
- ABIN2441994 reveals at least three additional non-specific bands of minor intensity in both the positive (wild type) and negative (dcl4-2) samples.
生效 #100563 (Western Blotting)![成功验证 '独立验证'标志]()
![成功验证 '独立验证'标志]() Validation ImagesFull Methods
Validation ImagesFull Methods -
-
状态
- Liquid
-
缓冲液
- 0.01 % Sodium azide
-
储存液
- Sodium azide
-
注意事项
- This product contains Sodium azide: a POISONOUS AND HAZARDOUS SUBSTANCE which should be handled by trained staff only.
-
储存条件
- -20 °C
-
-
- DCL4 (Dicer-Like Protein 4 (DCL4))
-
别名
- DCL4
-
分子量
- 191 kDa
-
UniProt
- P84634
抗原
-

 (1 validation)
(1 validation)



